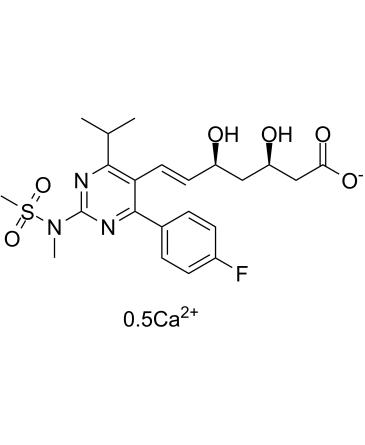
Rosuvastatin Calcium
| 瑞舒伐他汀钙 | Rosuvastatin Calcium | 147098-20-2 | In-Domus/CEP |
| RSM(Crystal) | 147118-40-9 | In-Domus | |
| RSM(Crude) | 147118-40-9 | In-Domus | |
| RSP | - | In-Domus | |
| RS-8 | 147118-36-3 | In-Domus | |
| RS-8-1 | 147118-37-4 | In-Domus | |
| TP-8 | 131466-61-0 | In-Domus | |
| TP-11 | 147118-35-2 | In-Domus | |
| TP-13 | 147118-39-6 | In-Domus | |
| RS-10 | 289042-12-2 | In-Domus | |
| RS-11 | 355806-00-7 | In-Domus |
Descriptio
Rosuvastatin Calcium (BANM, JAN, USAN) notum est Rosuvastatin in US. et concursus HMG-CoA inhibitoris reductase cum IC50 11 nM[1]. Rosuvastatin Calcium potenter praecludit a-go-ire humanum gene (hERG) currentem cum IC50 195 nM, repolarizationis cardiacis retardatum, ac per hoc longas actiones potentiales durationes (APDs) et intervalla QT intervalla (QTc) emendavit[2]. Rosuvastatin Calcium expressionem hERG maturae et commercii caloris incursus dapibus 70 (Hsp70) cum dapibus hERG minuit. Rosuvastatin Calcium efficacissimum est ad demissionem humilitatis densitatis lipoprotein (LDL) cholesterolum, triglycerides, et C-reactivum dapibus campestribus[3].
Praeterea notitia de nominibus medicamentorum conventionibus: Internationalis Nomina nonproprietaria.
Praecipua Notitia: Drus.com database in BETA emissio internationalis est. Hoc significat adhuc sub evolutione et indiligentias continere potest. Non destinatur substitutus peritiae ac iudicii tui medici, pharmacopolae vel alius sanitatis professionalis. Non debet indicare, cuiuslibet medicamentorum usus in quacumque regione tutus est, tibi idoneus vel efficax. Consule cum professionali sanitatis antequam ullum medicamentum accipias.
In US, Rosuvastatin (rosuvastatin systemica) membrum est status classium medicamentorum et adhibetur tractare Atherosclerosis, Maximum Cura, Maximum Cura - Familiale Heterozygoum, Maximum Cura - Familiale Homozygoum, Hyperlipoproteinemia, Hyperlipoproteinemia Type IIa - Elevatum LDL Type IIb - Elevatum LDL VLDL, Hyperlipoproteinemia Type III - Elevata beta-VLDL IDL, Hyperlipoproteinemia Type IV - Elevata VLDL, Hypertriglyceridemia et Praeventio Morborum cardiovascularium.
Background
A selectiva, auctorum inhibitor reductase HMG-CoA, id est etiam antilipemica.
Repono
| Pulvis | -20°C | III annos |
| 4°C | II annis | |
| In solvendo | -80°C | VI menses |
| -20°C | I mensis |
Chemical structure





Rogatio18Qualitas Constantia Aestimatio inceptis quae probaverunt4et6incepta approbantes.

Provectus qualitas internationalis administrationis systematis solidum fundamentum in venditionibus posuit.

Qualitas vigilantia percurrit totum vitae cyclum producti, ut qualitatem et therapeuticum effectum curet.

Negotiis professionales regulares turmas sustinet qualitatem postulatorum in applicatione et adnotatione.


Korea Countec Bottled Packaging linea


Taiwan CVC Bottled Packaging Line


Italia CAM Board Packaging Line

German Fette Compacting Machine

Iaponia Viswill Traba Detector

DCS Control Room





